Streamline Cell Line Expansion and Cryopreservation
Achieve Scalability and Traceability
Consistency and critical time constraints are just a couple of the most challenging aspects when handling cell lines manually. Automata's LINQ platform addresses these challenges by providing an end-to-end automation solution.
Maximize hands-off time and increase consistency with automation
LINQ enables the expansion and cryopreservation of large numbers of cell lines. Automate the entire process from cell seeding to cryopreservation.
Enhanced Data Integrity
The platform unifies data from multiple sources, providing comprehensive traceability essential for regulatory compliance.
Complete Workflow Integration
From cell seeding to master cell bank creation, LINQ supports a holistic processes within an adaptable system.
Reduced Manual Intervention
Automation minimizes human error and variability, leading to more reproducible and reliable outcomes.
Solutions
Cell Line Maintenance + Expansion + Cryopreservation
3D visualization of the work-cell highlighting key components for fully automated cell line maintenance. Wether you are working with suspension cells, adherent cells or organoids, this work-cell is capable of handling all your cell line maintenance needs, including but not limited to media change, cell count, passaging, cell imaging, cell suspension reformatting and cryopreservation.

Using LINQ Canvas for your Streamline Cell Line Expansion and Cryopreservation
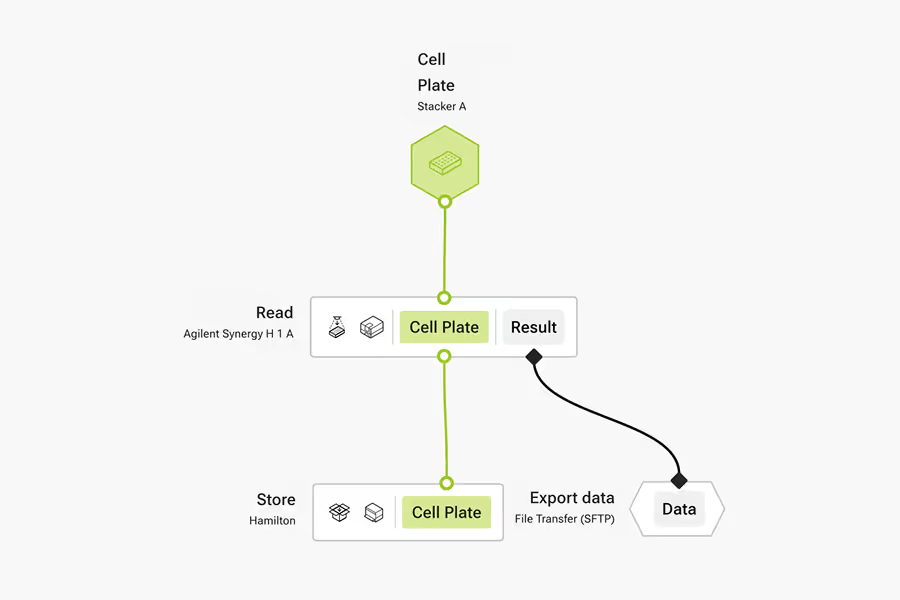
LINQ Canvas
Build workflows complete with data handling. Use LINQ Canvas to export images and cell health data seamlessly.

